The Evolution of Catalytic Converters: From Simplicity to High Efficiency
Uncover the remarkable journey of catalytic converters: from basic functionality to exceptional efficiency. Witness the transformative advancements in materials, coatings, and designs that have revolutionized emission control.



Suggested A-Premium Parts
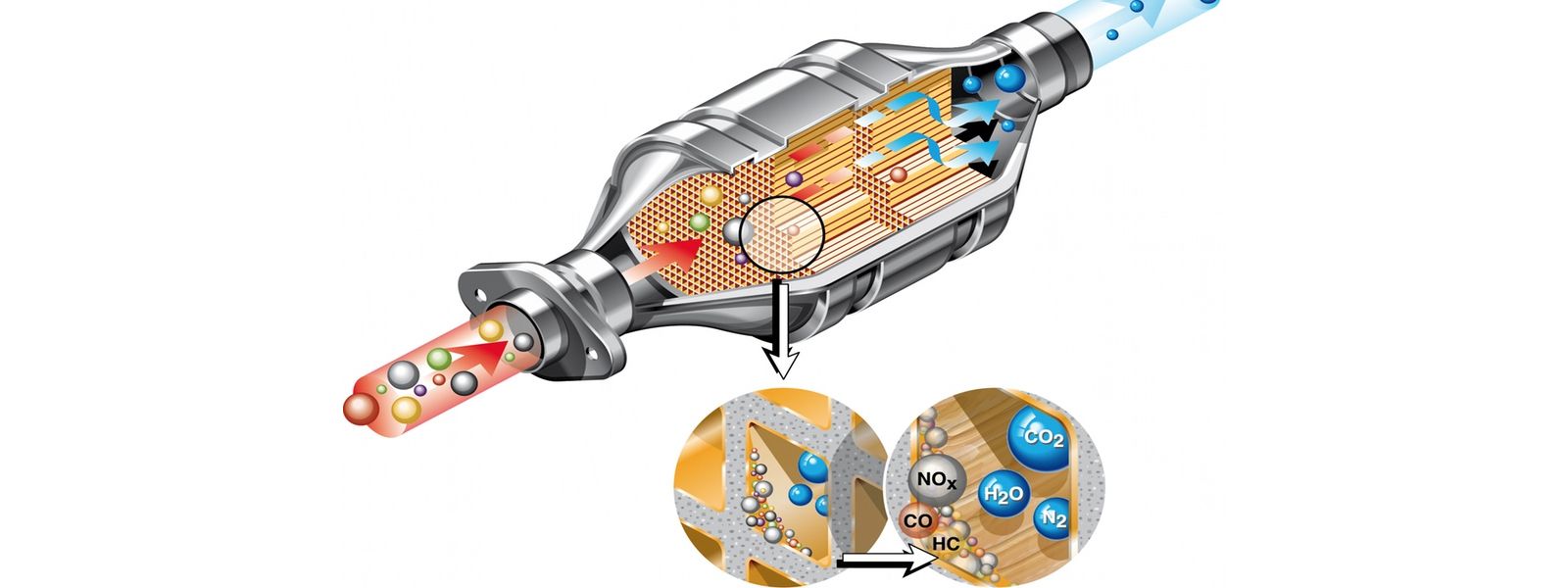
The catalytic converter is an important part of a car's exhaust system. Its main function is to convert toxic gases such as carbon monoxide, hydrocarbons, and nitrogen oxides in car exhaust into carbon dioxide, nitrogen, hydrogen, and water that are harmless to the human body. The development of catalytic converters has played a vital role in reducing vehicle emissions. With the continuous advancement of catalytic converter technology, its conversion efficiency continues to improve, and it can more effectively reduce the content of harmful substances in automobile exhaust. At the same time, with the development and application of new catalysts, the catalytic converter's ability to handle a variety of harmful gases has also been significantly improved. The evolution of catalytic converters not only helps automakers meet more stringent environmental regulations but also improves the fuel economy and power performance of vehicles.
Early Efforts in Emission Control: Two-Way Catalytic Converter
Early catalytic converters were mainly two-way catalytic converters, but due to their relatively simple design and function, there were some limitations and shortcomings.
The two-way catalytic converter is mainly based on the catalytic phenomenon in the chemical reaction process and converts harmful substances in automobile exhaust into harmless substances through the action of catalysts. These catalysts usually include precious metals such as platinum (Pt), rhodium (Rh) and palladium (Pd), etc.,
Allows oxidation of CO (carbon monoxide) to less-harmful CO2 (carbon dioxide)
Allows oxidation of HC (unburned hydrocarbons) to CO2 (carbon dioxide) and H2O (water)
In this design, exhaust gases are directed through a substrate containing the precious metals platinum and palladium, allowing a chemical reaction to occur. As the conversion process occurs, the exhaust gas temperature increases. Due to the intense heat generated by the process, the exhaust gas leaving the converter should be hotter than the gas entering the converter. This also explains why most equipment requires heat shields.
However, two-way catalytic converters have some limitations and disadvantages. First of all, due to the limited performance and life of the catalyst, its conversion efficiency is not high and it cannot completely convert all harmful substances. Secondly, the catalyst has high requirements for oil quality. If poor-quality fuel is used, the catalytic converter may fail or be damaged. In addition, early catalytic converters can usually only handle some harmful substances, and the treatment effect on nitrogen oxides (NOx) is not ideal. The ineffectiveness of controlling NOx (nitrogen oxides) led to the introduction of three-way converters.
Three-Way Plus Air Catalytic Converters
In the late 1970s and early 1980s, as the global awareness of environmental protection increased and vehicle exhaust emissions became increasingly prominent, more advanced vehicle emission control technologies began to be widely adopted in North America. During this period, three-way plus air converters (usually referring to the combination of a three-way catalytic converter and an air injection system) were widely used in vehicle emissions systems in North America to more effectively reduce harmful emissions in vehicle exhaust.
Allows reduction of NOx (nitrogen oxides) to N2 (nitrogen) and O2 (oxygen)
Allows oxidation of CO (carbon monoxide) to less-harmful CO2 (carbon dioxide)
Allows oxidation of HC (unburned hydrocarbons) to CO2 (carbon dioxide) and H2O (water)
There are two substrates inside the converter. The front is coated with the precious metal rhodium, which is used to reduce NOx emissions into simple N2 and O2. This process works best when a small amount of oxygen is present (a rich mixture). That's why it's located upstream of the air tube. Because the high-concentration mixed gas contains high amounts of HC and CO, the air pump and tubes will add oxygen to the mixed gas before being transported to the next substrate. The second layer of substrate is coated with precious metals Pd and Pt, which can effectively convert harmful gases such as HC and CO into low-toxic gases CO2 and H2O. This system was not very efficient and was phased out in the early 1980s when the current three-way converter was introduced.
Breakthrough and Innovation: Three-Way Catalytic Converter
Since 1981, three-way catalytic converters have served as a core component of vehicle emissions control systems in North America and many other countries. The introduction of this technology marks a new stage in vehicle exhaust gas treatment, which plays a vital role in reducing air pollution and improving environmental quality.
Allows reduction of NOx (nitrogen oxides) to N2 (nitrogen) and O2 (oxygen)
Allows oxidation of CO (carbon monoxide) to less-harmful CO2 (carbon dioxide)
Allows oxidation of HC (unburned hydrocarbons) to CO2 (carbon dioxide) and H2O (water)
Air-free three-way catalytic converters have the unique ability to store and release O2. When the oxygen content in the exhaust gas is high, the catalyst can absorb and store excess O2; when the O2 content in the exhaust gas is low, the catalyst can release the stored O2, providing the necessary conditions for the oxidation reaction. In addition, to further improve converter performance, modern vehicle emission control systems incorporate O2 monitoring and control systems. The system uses one or more oxygen sensors to oscillate the fuel mixture between lean and rich states. This oscillation, combined with O2 storage and release at the catalyst surface, can minimize all three emissions. Three-way catalytic converters are used in conjunction with the OBDII diagnostic systems found on today's vehicles. The system alerts the driver when the converter is not operating at maximum efficiency.
Ongoing Challenges for Catalytic Converter
Catalytic converter technology plays a central role in automobile emission control systems. Although it has achieved remarkable results in reducing the emission of harmful substances in exhaust gases, it still faces a series of ongoing challenges. These challenges mainly include:
Catalyst service life: The activity, stability and service life of the catalyst are important factors that determine its working efficiency. As environmental protection becomes increasingly stringent, higher requirements are placed on catalyst performance. The development of efficient and stable catalysts is an important challenge currently faced.
High-temperature stability: Catalytic converters need to work in high-temperature environments, and high temperatures may cause catalyst sintering and deactivation. Therefore, it is an important technical problem to improve the high-temperature stability of the catalytic converter and ensure that it can still maintain efficient conversion at high temperatures.
Cost issue: High-performance catalysts often use precious metals as active ingredients, resulting in higher manufacturing costs for catalytic converters. How to reduce manufacturing costs while maintaining performance is an economic challenge that catalytic converter technology needs to solve.
Adapt to new fuels and engines: With the development of automotive technology, new fuels and engines continue to emerge, placing higher requirements on the performance of catalytic converters. How to adapt to the needs of different fuels and engines is a challenge that catalytic converter technology needs to address.
In summary, catalytic converter technology faces many challenges in its development, and continuous technological innovation research and development are required to address these challenges.